Navigating Compliance with Expertise and Precision
At CSA Quality Consulting Pvt. Ltd., we offer comprehensive Regulatory Affairs services designed to guide your products through the complex landscape of global regulatory requirements. Our team of experienced professionals ensures that your clinical trials and product developments comply with the necessary regulations, facilitating smooth market access and minimizing delays.
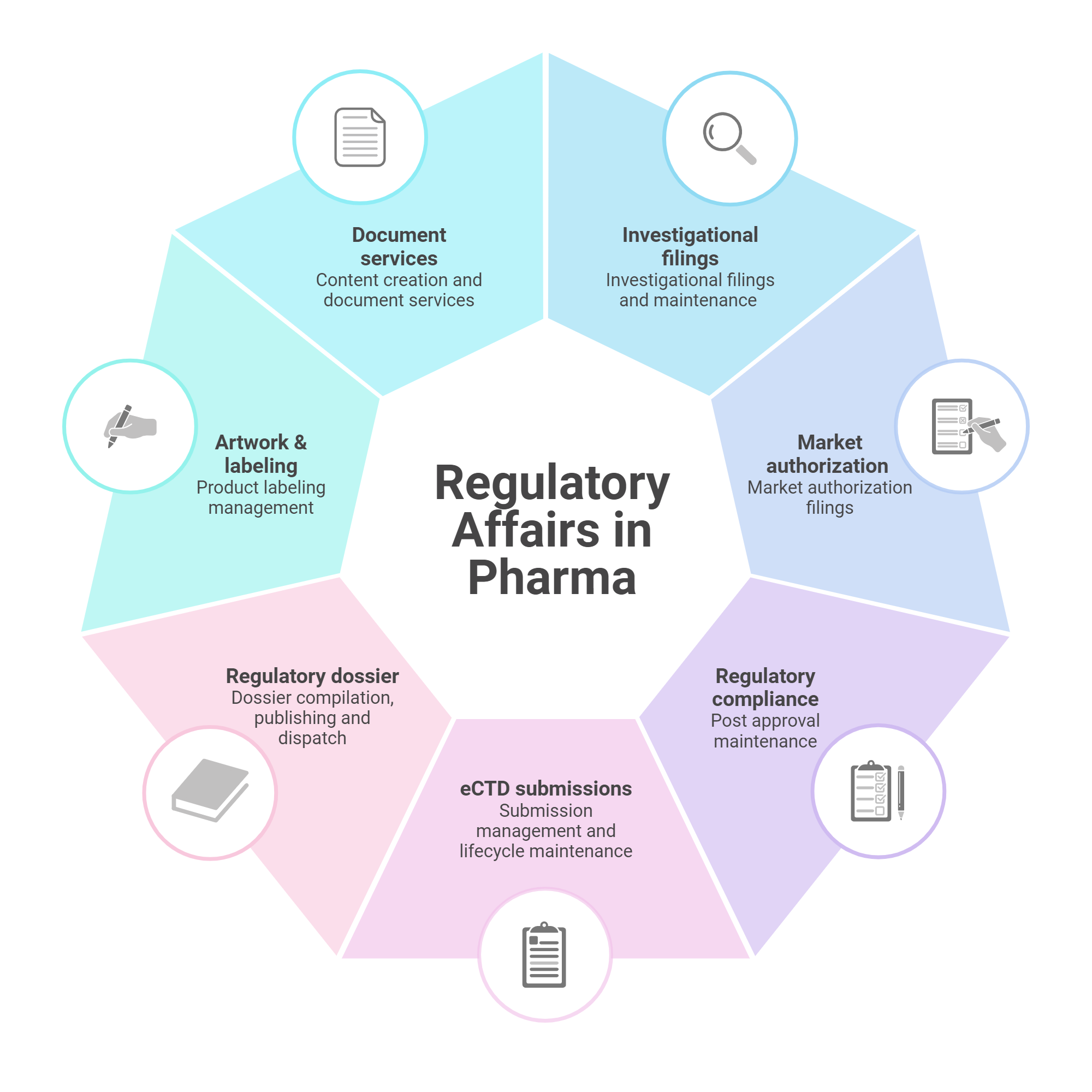
Our Regulatory Affairs Services Include
Regulatory Strategy Development
Crafting tailored strategies to navigate regulatory pathways efficiently.
Regulatory Submissions
Preparing and submitting dossiers for approvals from regulatory bodies such as the CDSCO, EMA, and FDA.
Compliance Audits
Conducting internal audits to ensure adherence to Good Clinical Practice (GCP) and other regulatory standards.
Labeling and Documentation Support
Assisting with the preparation and review of labels and other essential documents to meet regulatory requirements.
Post-Marketing Surveillance
Monitoring and reporting on the safety and efficacy of products post-launch to ensure ongoing compliance.
Why Choose CSA Quality Consulting?
Expertise Across Therapeutic Areas
Our team has extensive experience across various therapeutic areas, ensuring specialized support for your projects.
Global Regulatory Knowledge
We stay abreast of global regulatory changes, providing you with up-to-date guidance.
Tailored Solutions
We offer customized services to meet the unique needs of your clinical trials and product developments.
Partner with CSA Quality Consulting Pvt. Ltd. to navigate the regulatory landscape with confidence and ensure the success of your products in the global market.
